San Leandro, CA – VIVIO, the leading drug outcomes company, proudly announces third-party validation of specialty drug savings delivered for VIVIO customers. Validation Institute independently certified a self-funded employer with 27,000 covered lives that spent 39% less in their first year with VIVIO than they did in the previous year with a Big-3 PBM.
The analysis compared an employer’s specialty drug costs in 2021 with a traditional PBM to that same employer’s specialty drug costs in 2022 managed by VIVIO.
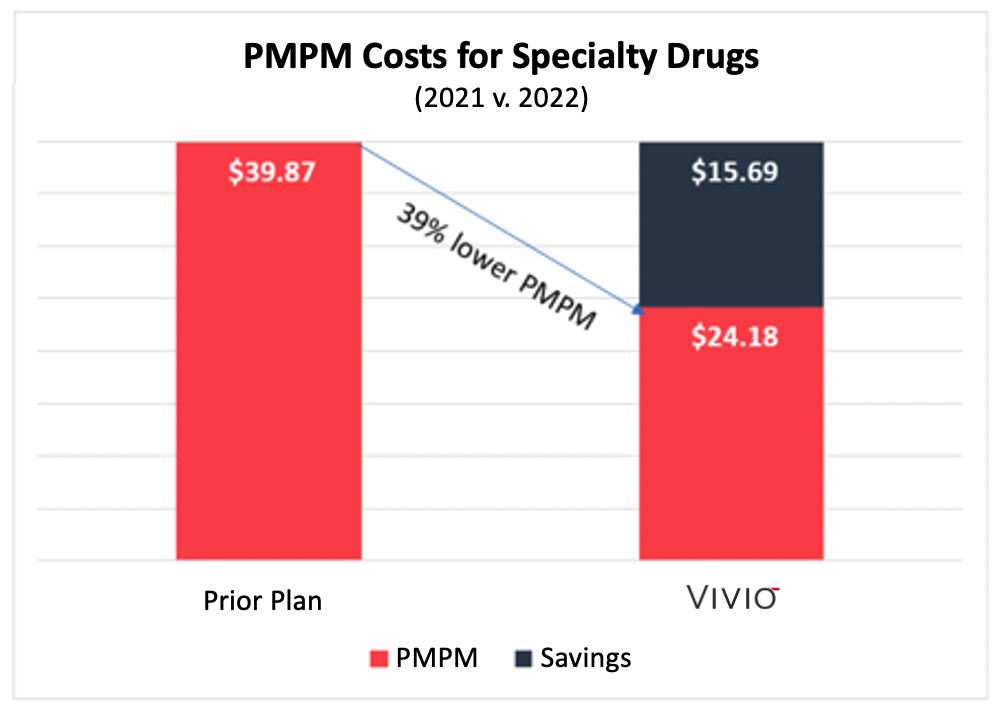
This chart summarizes the Per Member Per Month (“PMPM”) costs for the employer before VIVIO and the following year with VIVIO. VIVIO’s PMPM costs were 39% lower than under the prior plan.
While PBM models focus on formularies that exclude drugs and limit drug access, VIVIO takes the opposite approach of personalizing drug therapies for each patient without formulary restrictions.
“As an independent, objective third-party organization, Validation Institute provides unbiased, data-driven insights on healthcare programs. The goal is to put more purchasing power into buyers’ hands,” said Benny DiCecca, Validation Institute CEO & President. “Our team found that the VIVIO program significantly reduces specialty drug costs.”
“We are delighted to have received independent third-party validation of the VIVIO program,” said Chris Crawford, VIVIO’s Chief Growth Officer. “We have taken a different approach than the traditional PBM model, which is proven to deliver better health outcomes for the members we serve and lower costs for our employer customers.”
About VIVIO
We prioritize health outcomes by fixing the following system problems: identifying expensive drug therapies that don’t work even though they have FDA approval; not knowing if a member is responding adequately to the therapy; doctors not reading the clinical trials themselves; the arbitrary line between pharmacy and medical benefit; and, egregious supply chain waste. VIVIO uses clinical trial, patient, and financial data to drive better health outcomes while eliminating wasted spending. VIVIO Precision Care™ plugs into an employer’s current carriers and PBMs. In 2021, VIVIO customers spent 67% less on specialty drugs than the national benchmark.
About Validation Institute
Validation Institute is a professional community that advocates for organizations and approaches that deliver better health value and stronger health outcomes at lower cost. We connect, train, and certify health care purchasers, and we validate and connect providers delivering superior results. Founded in 2014, the mission of the organization has consistently been to help provide transparency to buyers of health care. To learn more about Validation Institute, visit validationinstitute.com
Media contact:
T. J. Tedesco
[email protected]
For PDF of this news release, click here.
Apparently the often-low standards for the FDA aren’t low enough. Now we just move trials to poor countries and use control arms that we wouldn’t approve here. VIVIO team member Bishal Gyawali writes about this in the Lancet. Look at the data yourselves and stop listening to the ‘experts’ who are paid to say ‘yes.’
VIVIO clinical team member, Bishal Gyawali, MD, PhD, is part of the team that develops the WHO Essential Medicines List (EML) identifies priority medicines that are most important to public health. Over time, the EML has included an increasing number of cancer medicines. See the details of their ground breaking work.
VIVIO clinical team member, Daniel Goldstein is referenced in Peter Bach’s scathing commentary in Health Affairs on the waste being driven by vial size games played by Pharma. The original article describes how Merck’s increase of vial size has led to billions of dollars of Keytruda being thrown away.
VIVIO clinical team member, Bishal Gyawali, MD, PhD, on why financial toxicity deserves more attention. “The more lung cancer patients had to pay out of pocket for treatment, the less likely they were to adhere to, or continue, therapy, reported researchers, who also found an indication of resulting less-than-ideal survival odds.”
https://www.medpagetoday.com/reading-room/asco/lung-cancer/92003
Bishal Gyawali, MD, PhD, VIVIO clinical team member, interviewed. According to Gyawali, “what’s the point in having a drug that does not reach patients because of unaffordability? Both high cost and low probability of clinical benefit should make us question the value of a drug, not hail it as a game-changer.”
https://journals.lww.com/oncology-times/blog/3questionson/pages/post.aspx?PostID=149
Pramod John, PhD, CEO of VIVIO Health, discusses a recent presentation where he spoke about using real-world data to drive better outcomes and lower costs, as well as weighs the benefits of value-based care.
https://www.managedhealthcareconnect.com/articles/using-real-world-data-right-way

